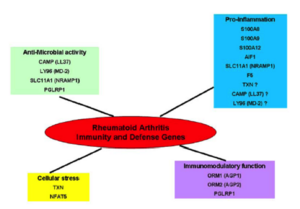
Differentially expressed genes in RA
From 2007, we have started the characterization of RA biomarkers with transcriptome analysis. We have benefited of RA cohort collected by Dr. P. Hilliquin and prepared for storage and sending by Dr Quillet and Dr Lemaire (GenHotel –Rheumatology & Biology Departments, CHSF Corbeil-Essonne). We have then extracted total RNA from blood samples of RA patients (cases). Through another in-house project, we obtained samples for healthy people (controls) (Dr. L Jacq, GenHotel – Cardiology Department, CHSF Corbeil-Essonne).
Our first goal was to measure expression of RA candidate genes by quantitative PCR (qPCR) and to study relation between level of expression and polymorphism of the candidate gene. We have then studied PRKCH gene (Teixeira et al., 2008a) and CASP7 gene, coding Caspase 7. Caspases are proteases involved in apoptosis mechanisms, which could be deregulated and contributed then to synovioycytes proliferation or to osteoblasts destruction, two physiopathological characteristics in RA. Measurement of expression level for alpha and beta isoforms of Caspase 7 show a significant decrease in cases in comparison with levels in controls, this one being more significant for alpha isoform. Alpha/beta ratio of expression level is then significantly decreased in cases in comparison with controls, suggesting a lower apoptotic activity related to alpha active form of caspase in RA (Teixeira et al., 2008b).
To extensively benefit of collected samples, we have then developed a collaboration with Dr. R. Olaso (Plateforme de Transcriptome, Centre National de Génotypage, Institut de Génomique, CEA, Evry). We performed whole transcriptome analysis using Illumina microarray technology on peripheral blood mononuclear cells (PBMCs) from RA cases and controls. We identified a remarkably elevated expression of a spectrum of genes involved in Immunity and Defence in PBMCs of RA cases compared to controls. This result is confirmed by GO analysis, suggesting that these genes could be activated systemically in RA (see figure; (Teixeira et al., 2009); PhD : https://www.theses.fr/147412374).
Among the genes showing the highest expression level change in this study, we have then carried out analysis of those located in a genomic region without copy number variation (CNV) known. Our goal was to analyze relation between expression and SNP and then identify expression Quantitative Trait Loci (eQTL) specific of RA. We have selected genes for which tagSNPs were suitable for our study. We then identify a suggested association of a SNP located in PGLYRP1 with RA but this preliminary result was not replicated in an extended sample of families (Fodil et al., 2015, co-supervising of PhD with Pr A Boudjema, USTO, Oran, Algeria, http://www.theses.fr/2015EVRY0017). Such study, focused on the relation between polymorphisms and expression level in complex diseases, constitutes a field of investigation significant in the determination of regulation regions associated to a specific phenotype.
Differentially expressed genes in pre-RA states
RA can be detected years before the first symptoms of the disease, with the development of a systemic autoimmunity. Indeed, auto-antibodies such as the rheumatoid factor and anti-citrullinated peptides antibodies (ACPAs), precede the clinical disease by a median period of at least 5 years.
Rheumatoid arthritis (RA), once fully developed, is difficult to treat and generally requires lifelong therapy. Treatments in the very early phases of the disease, or ideally before the clinical onset of the disease (= pre-clinical phases), are potentially curative. Several prevention trials for RA are ongoing and may lead to screening and preventive strategies for RA, much as controlling hypertension and reducing high cholesterol is helping to reduce the risk of cardiovascular diseases (Finckh A. et al. 2014) However, before preventing RA can become a reality, the precision of diagnosing preclinical RA will need to be improved. While the hereditability of RA is well established and preclinical stages have been identified, it is currently still impossible to provide patients with an individualized estimate of RA risk. Thus the precise diagnosis of pre-clinical RA has become a major scientific question.
Our work hypothesis is that the asymptomatic, pre-clinical phase of RA can be adequately identified by a combination of biologic markers and clinical risk factors. And the characterization of specific regulation profiles and biological abnormalities lead to the identification of new biomarkers present before the first symptoms appear and predictive of RA onset. Our objective is to establish the mechanisms of disease initiation: identify gene regulations in individuals who develop the disease within a year (pre-RA samples) and specific sequence of biological abnormalities leading to the development of disease. We then will be able to characterize biomarkers predictive of RA onset within one year, to be followed-up for evaluation of the diagnostic value for prognostic or treatment response and to characterize individuals at very high risk of developing RA.
This project is based on the SCREEN-RA cohort (www.arthritis-checkup.ch) aims to develop and evaluate a screening strategy for the development of RA in individuals genetically at risk, namely first degree relatives of patients with autoimmune diseases. The cohort was established by Pr. Axel Finckh,(HU Geneva, Switzerland), with the help of a previous SNSF grant (SNSF N° 32003B_120639). Since 2010, over 1300 first degree relatives of RA patients have enrolled, given informed consent, answered detailed epidemiological questionnaires and provided biologic samples (serum, DNA, RNA, stool). The study continues to enrol around 200 new participants per year. Only individuals without clinical evidence of RA are enrolled and followed-up yearly to assess incident arthritis or other phases of impending RA. Our project is also based on a similar cohort developed in France by Pr. F Cornélis.
A preliminary study has been performed with transcriptome analysis through array technology with the collaboration of Dr. R Olaso from CNRGH (Institut François Jacob, CEA/DSV, Evry). Analyses of results are in progress and benefit of the collaboration of Dr. C Dalmasso from LaMME laboratory (UMR8071 CNRS, Evry University).